 Several relationships govern geometry including bonding, electron. a - Trigonal pyramidal, linear b - Tetrahedral, trigonal planar c - Trigonal planar, trigonal planar d - Tetrahedral, bent e - Trigonal pyramidal, seesaw 2) Of the molecules below, only is polar. a) The electron-domain geometry is tetrahedral. Molecular geometry is the spatial arrangements of atoms that are bonded in a molecule. 1) The electron domain and molecular geometry of BrO2- is. 
Choose the answer which gives the electron-domain geometry and molecular geometry which are both correct for TeF4. The electron-pair geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be. In molecules with more than three atoms, there are many more possible geometries. 9.1: Molecular Geometry 9. The electron domain and molecular geometry of SO3 are: a. XeF4 has sp3 d2-hybridized Xe-atom having two lone pair of electrons. If the B atoms are halogens, each will have three. The molecular and domain geometry of XeF4 respectively are square planar and octahedral. 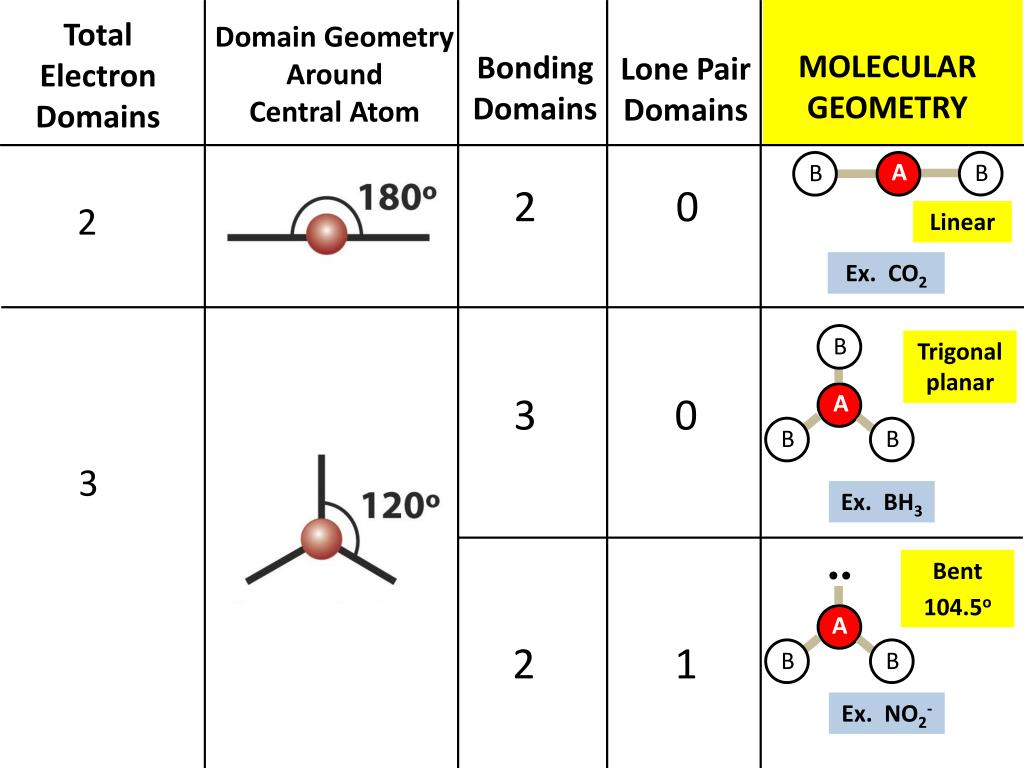
However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. With five bonding and one nonbonding electron domains, the molecule has octahedral domain geometry. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. 
Explanation: Electronic geometry takes into account the electron pairs that are not participating in bonding, and the electron cloud density. The molecular geometry gives water a bent shape. A polyatomic molecule contains more than two atoms. Molecular structure describes the location of the atoms, not the electrons. The electronic geometry gives water a tetrahedral shape. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
